Prices & Payments
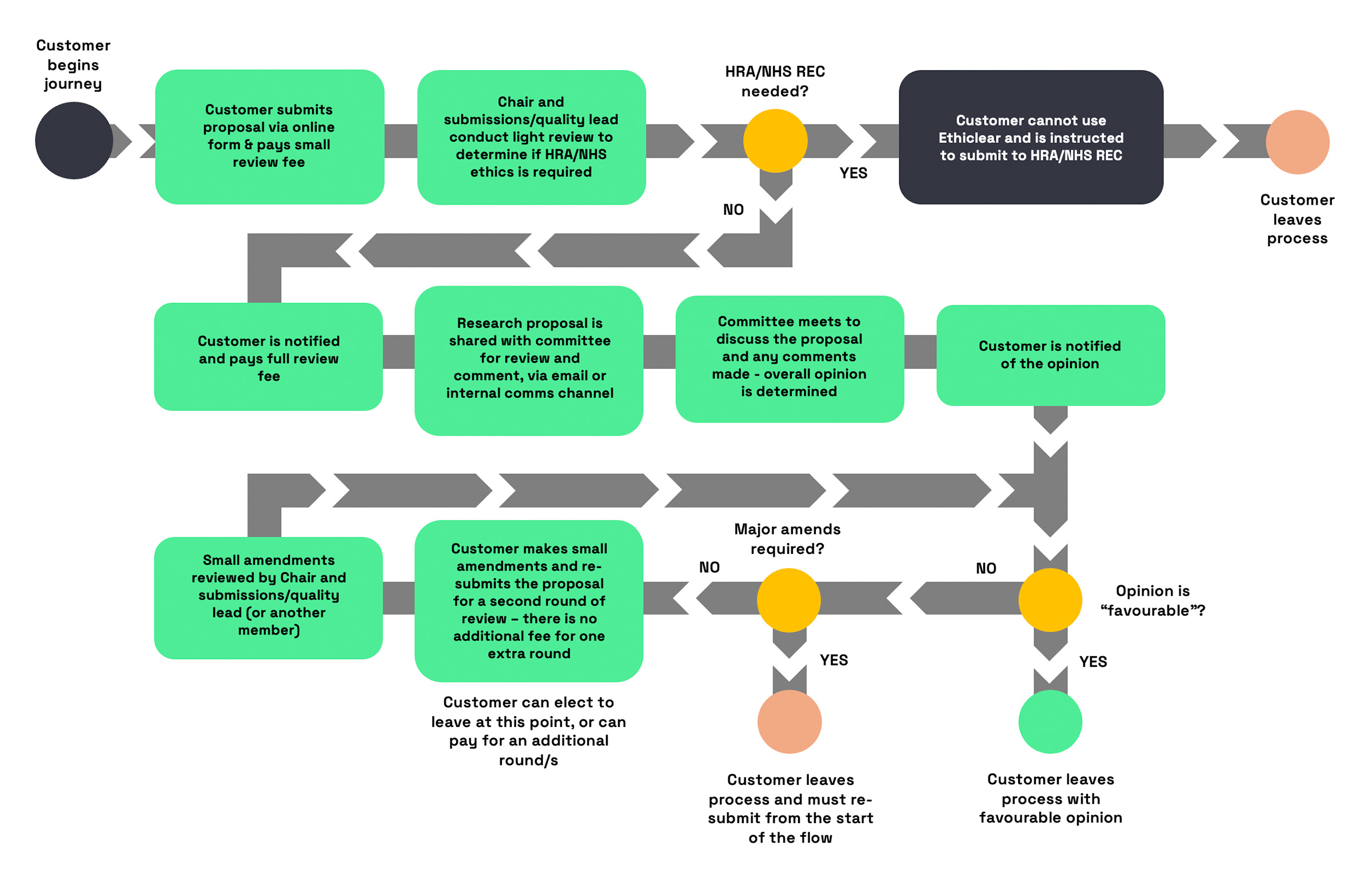
The initial (“small”) review fee is charged at
£100.00, and includes a check over the material submitted to ensure that Ethiclear is able and allowed to review the project proposal. At this stage, Ethiclear administration and quality assurance members will also work to ensure that all required information has been provided, and may request additional information or documentation from the customer before the project can progress to full review.
If and when the project can progress to full review, an additional fee of
£1,000.00 is charged in order to provide the following:
>>
Full review by the committee
>>
Meeting and discussion time>>
Report generation>>
One round of
minor amendment review
All prices quoted are
exclusive of VAT.
All payments are
non-refundable and must be made (or invoice raised with 30-day terms) before the review can begin.
Payments can be made via bank transfer or via credit/debit card.
Discretionary discounts may be provided to non-profit organisations and charities. Customers may wish to contact
hello@ethiclear.com to discuss their needs in such cases. Upon research project submission, where such a discount is requested, Ethiclear administration will reach out to the customer to understand needs.
Sponsor ResponsibilitiesFor the purposes of this policy, sponsor refers to the study sponsor of the research proposal or, if different, the entity submitting the project to Ethiclear.
Regulatory Compliance:
The responsibility to determine whether
Health Research Authority (HRA) / NHS Research Ethics Committee approval is required lies entirely with the study sponsor. Sponsors (and project submitters) must use relevant HRA decision tools and seek appropriate statutory approvals where required. This can be found here:
https://www.hra-decisiontools.org.uk/ethics/
The responsibility to seek appropriate ethics review, and any other regulatory approvals, lies purely with the sponsor.
Information Accuracy:
Sponsors must provide complete and accurate information for review. Any material changes to study protocols must be reported immediately, and all parties understand that the ERB's opinion is based solely on information provided at the time of review, and is based on information limited only to that submitted.
If the subsequently-run study differs in any way from the information submitted to Ethiclear, the outcome from the ethical review is no longer valid.
Independence & Conflicts of InterestDeclaration Requirements:
All committee members must internally declare any potential conflicts of interest upon receipt of project proposal information.
Furthermore, any financial, personal, or professional interests that may affect impartiality, in addition to any relationships (personal, professional or otherwise) with study sponsors, investigators or competitors, must also be declared.
Such declarations, at the Chair and/or Ethiclear administration’s discretion, may result in the member being excluded from the review of that research project.
Members may also recuse themselves from the review of any project for any reason they may have; whether personal or professional. In such cases, whilst a justification for such a recusal is useful to know, there is no obligation for the member to reveal this.
Conflict Management:
Members with declared conflicts may be required to withdraw from relevant discussions and decisions related to the relevant research project. However, this will not in itself affect their involvement in other projects.
The ERB chair and/or Ethiclear administration will determine appropriate conflict management measures in-line with the type and severity of the declaration. Where the member is still permitted to conduct the review, the declaration and resultant decisions must be logged with justifications provided.
If a member declares or is found to be in severe or repeated conflict, the member may be excluded from specific future projects automatically.
The ERB Chair or Ethiclear administration may decide to exclude committee members from a review/project if it is felt that there is potential risk or conflict of interest or other involved risk.
Independence Requirement:
Committee members must act independently of personal, professional, or commercial interests. Members are reimbursed for their time irrespective of the ultimate ethical opinion determined.
Members must maintain objectivity when also working for competitors or related organisations. All members sign and agree to non-disclosure agreements (NDAs) when joining the Ethiclear ERB, and thus have legal obligations to maintain full confidentiality with regard to all and any work conducted as part of the Ethiclear ERB.
Participant welfare and ethical considerations are paramount, and must be considered above all other concerns.
Data Protection & ConfidentialityInformation Security:
All review materials are confidential and must be handled according to company data protection and security policies. All committee members (including administrative staff) must sign appropriate contracts (including Non-Disclosure Agreement (NDAs) clauses or schedules) when starting work with or on the Ethiclear ERB.
Electronic documents are stored securely and accessed only by authorised personnel. Company policy dictates that sensible security practices are adhered to, including the use of disk encryption, secure storage solutions, use of antivirus/anti-malware solutions, and secure workflows.
Data Retention and Deletion:
All sponsor-provided materials (information submitted to Ethiclear) must be securely deleted from committee member devices within seven days of final report issuance.
Some or all materials and documentation may be retained indefinitely by Ethiclear administration (excluding non-relevant personal data, where there is no legal basis to do so). For example, this may enable Ethiclear to conduct follow-up reviews of related or similar studies, or to receive updates on the progress or completion of previously-reviewed projects. The sponsor may request that such materials are removed from Ethiclear systems after the review completes.
Any personal data will be processed in accordance with UK GDPR requirements. Please refer to the Ethiclear Privacy Policy for more information.
Ethiclear ERB committee members are not employees of Seastorm Limited. Whilst training processes, contracts and NDAs, and security controls are in-place, committee members work and operate and conduct their reviews independently.
Reporting & DocumentationFinal Report Requirements:
Each review will produce a final report containing:
>> Overall ethical opinion (Favourable/Non-Favourable/Major Non-Favourable)
>> Rationale for the decision and any related comments
>> Specific recommendations or required modifications
>> Committee member information from those participating in the decision
>> Review date and a unique reference number.
Report Distribution:
Final reports will be provided to study sponsors within one business day of the committee meeting in which the relevant research project is discussed. Internal copies of such reports are retained by Ethiclear administration for audit and quality assurance purposes.
In cases of non-favourable opinion, amendments made based on Ethiclear ERB comments and/or recommendations can be re-submitted via email. A new report will be issued in relation to the amended submission.
Committee decisions are final and binding.
Committee Member ObligationsHandbook and Training Compliance:
All committee members must adhere to all provisions in the Committee Member Handbook, complete and refresh mandatory training programmes, maintain any required professional or continuing education requirements, and follow all relevant company policies and procedures.
Non-Disclosure Obligations:
All committee members must sign and comply with Non-Disclosure Agreements, maintain strict confidentiality of all review materials, not discuss review details outside official committee business (including with friends and family), and secure all confidential documents appropriately and in-line with Handbook and policy guidance and requirements.
Disciplinary Measures:
Breach of committee member obligations may result in any or all of formal warning, mandatory retraining, temporary suspension from committee duties, permanent removal from the committee, or legal action for serious breaches of confidentiality.
Public AccountabilityCommittee Member Disclosure:
Names and short biographies of current committee members will be published on the company website and made available on other relevant material. Relevant member qualifications and expertise areas will also be publicly available.
Contact information for the ERB administration is publicly available.
Transparency Measures:
Annual reports summarising ERB activities (anonymised) may be published. Committee meeting schedules (if set) will be made available to potential applicants.
This Ethics Policy is made publicly available on the Ethiclear website.
Balancing Innovation & EthicsProportionate Review:
The ERB commits to applying proportionate ethical oversight appropriate to study risk levels and supporting legitimate scientific innovation while maintaining ethical standards.
Members will individually and collectively provide constructive feedback to improve study design where possible and facilitate timely review processes to support research advancement.
Continuous Improvement:
Ethiclear welcomes feedback from sponsors, researchers, and committee members. It is Ethiclear’s collective mission to make ethical review accessible, safe and fair to all organisations conducting good research.
Ethiclear will continually benchmark itself against best practices in research ethics review, following NHS/HRA guidelines, in addition to relevant guidelines published by other regulatory bodies, and adjust its processes and practices accordingly.
Policy Review:
This policy will be reviewed annually (or on-demand or in response to business need or external factors, including regulatory, and internal or external feedback), with updates made as/when relevant.
Annual reviews and audits will also be made against committee processes and decisions and customer feedback to ensure maintained quality assurance.
Annual training reviews will be made for all committee members, highlighting and remediating any gaps in professional, operational or security knowledge and skills.